Post hoc subgroup analysis
References:
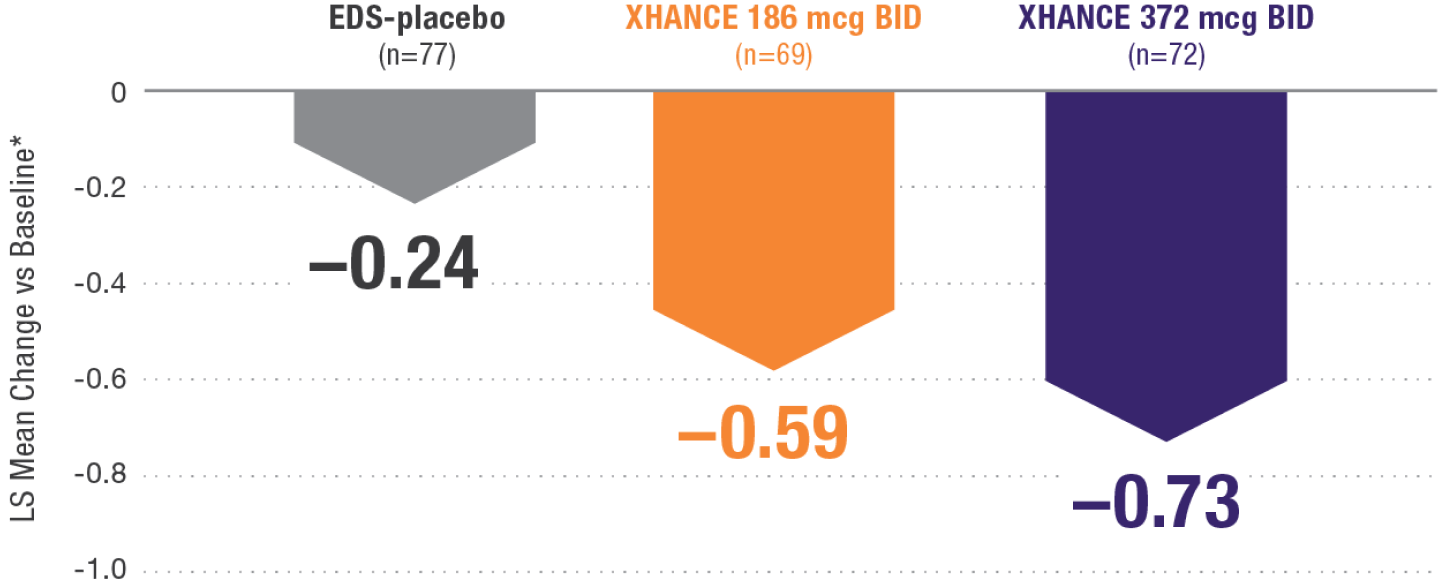
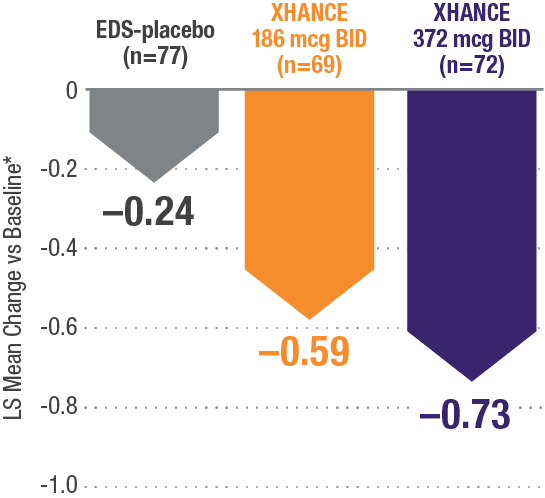
CONGESTION/OBSTRUCTION REDUCTION IN PATIENTS PREVIOUSLY USING STANDARD NASAL STEROIDS1,2
Reductions in congestion/obstruction at Week 4 in the subgroup of patients reporting standard nasal steroid use prior to study entry1,2
In a subgroup of patients reporting use of a standard nasal steroid within 30 days of the screening visit1:
- Patients reported a mean treatment duration of 3 years with a standard nasal steroid
- The improvements in this subgroup were similar in magnitude to improvements in overall study population
Post hoc analysis of NAVIGATE I and II Pooled subgroup data (n=218)


*Least-squares mean change from baseline in patient-reported AM instantaneous daily scores for nasal symptoms on a scale from 0-3 (0=none, 1=mild, 2=moderate, 3=severe).1 BID=twice daily.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS:
- Local Nasal Effects: epistaxis, erosion, ulceration, septal perforation, Candida albicans infection, and impaired wound healing. Monitor patients periodically for signs of possible changes on the nasal mucosa. Avoid use in patients with recent nasal ulcerations, nasal surgery, or nasal trauma until healing has occurred.
- Senior BA, Schlosser RJ, Bosso J, Soler ZM. Efficacy of the exhalation delivery system with fluticasone in patients who remain symptomatic on standard nasal steroid sprays. Int Forum Allergy Rhinol. 2021;11(5):837-845.
- Full Prescribing Information for XHANCE (fluticasone propionate). OptiNose US, Inc.; 2023.